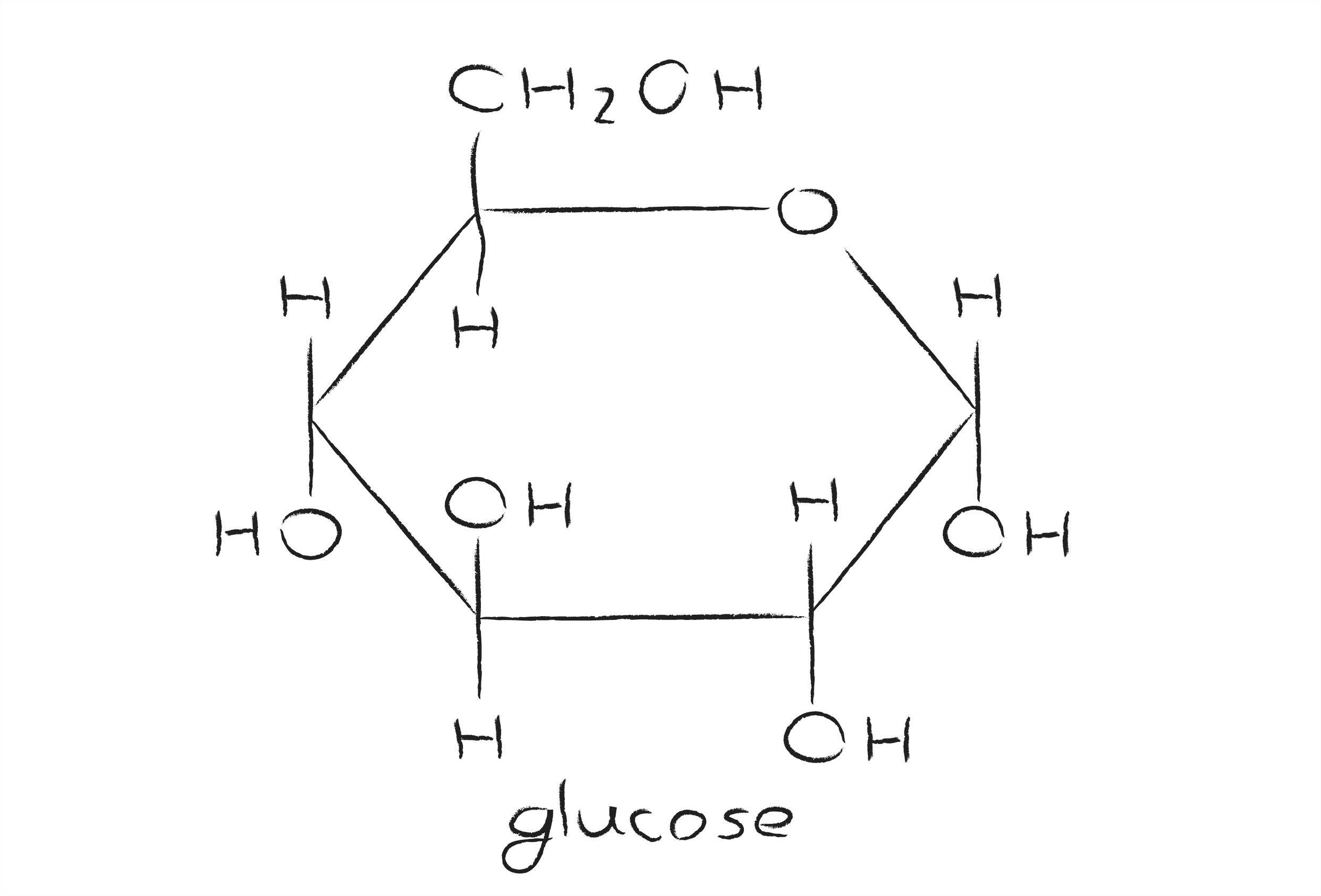
1 mole of glucose has a mass (its gram formula weight or molar mass) which is the sum of the gram atomic weights of all of t he constituent atoms. Mass of 1 mole of glucose, C 6 H 12 O 6 = (6 × 12.01 + 12 × 1.01 + 6 × 16.00) g = 180.18 g (using atomic weight data to 2 decimals). Given: Mass of urea = 250 g, cost for 250 g glucose = Rs 40, molecular formula of glucose = C 6 H 12 O 6 To find: Cost per mole of glucose Calculation: Molecular formula of glucose is (C 6 H 12 O 6). Molecular mass of glucose = (6 × Average atomic mass of C) + (12 × Average atomic mass of H) + (6 × Average atomic mass of O).
- Glucose Mw
- Atomic Mass Of Glucose C6h12o6
- Molar Mass Of Sugar
- Atomic Mass Of Simple Sugar Glucose
- Molar Mass Of White Sugar
- Glucose Moles
Formula Weight: sum of masses of all atoms in a given formula or compound
Formula Weight = (# atoms of A) (atomic mass of A) + (# atoms of B) (atomic mass B) …
Example: What is the formula weight of glucose: C6H12O6?
Steps to Solve:
1. List each element and the # of atoms of that element are in the formula
C: (6 atoms of C)
H: (12 atoms of H)
O: (6 atoms of O)
2. Multiply the number of atoms of each element by that element’s respective mass (from the periodic table) to determine the TOTAL mass contributed to the formula from each element in small “chunks:”
Federal Excise Tax (FET) — a tax imposed on premium payments to offshore insurers: 4 percent on direct premiums and 1 percent on reinsurance premiums. It was once relaxed for premium payments to most offshore insurers/reinsurers in Barbados and Bermuda, but not for long. In fet. A frozen embryo transfer (FET) is a type of IVF treatment where a cryopreserved embryo created in a full IVF cycle is thawed and transferred to a uterus. FET typically uses “extra” embryos a couple has from a previous conventional IVF cycle. A cryopreserved embryo can also be a donor embryo. FET-Self Bias circuit. This is the most common method for biasing a JFET. Self-bias circuit for N-channel JFET is shown in figure. Since no gate current flows through the reverse-biased gate-source, the gate current I G = 0 and, therefore, v G = i G R G = 0. With a drain current I D the voltage at the S is V s = I D R s. The gate-source voltage. FET is an acronym for federal excise tax. It refers to the tax imposed by the federal government on tires used on the road with a maximum load capacity greater then 3500 pounds. Generally this applies to medium truck tires and heavy duty trailer tires. Do I pay FET on farm and forestry tires? IN-FET is the acronym of a European scientific project consortium, funded by the European Commission during 2020-2023. It stands for Ionic Neuromodulation For Epilepsy Treatment and brings together engineers, biologists, physicists, and neuroscientists to tackle the hard problem of.
Glucose Mw
C: (6 atoms of C) x (12 g) = 72 g
H: (12 atoms of H) x (1 g) = 12 g
O: (6 atoms of O) x (16 g) = 96 g
3. Add all accumulated masses for each element in the formula to calculate the formula weight:
72 g + 12 g + 96 g = 180 g
So formula weight of glucose = 180 g
Atomic Mass Of Glucose C6h12o6
Let’s try one more…
Example: What is the formula weight of aluminum sulfate? Al2(SO4)3 [Ans: 342 g]
Solution:
Al: 2 atoms x 27 g = 54 g
S: 3 atoms x 32 g = 96 g
O: 12 atoms x 16 g = 192 g
S: 3 atoms x 32 g = 96 g
O: 12 atoms x 16 g = 192 g
Molar Mass Of Sugar
54 + 96 g + 192 g = 342g Atoms are neutral because the number of.
Atomic Mass Of Simple Sugar Glucose
FYI: Amu and Grams
Molar Mass Of White Sugar
While grams are typically used to calculate formula weight because they are more practice, did you know that the first unit used to denote atomic mass–although now arbitrary—was the atomic mass unit. You may see the “amu” unit used for mass in textbooks, but not to worry, there is no need to convert from one to the other. Luckily, they mean the same thing! So 1 amu = 1 gram.
Glucose Moles
Why? Every amu is the same as a gram thanks to a clever notion: that 1 amu is equivalent to the 1/12 of the mass of a carbon atom (12 on the periodic table) in amu. So 1/12 x 12=1. That is how we attain the 1:1 ratio.
